08-03-2015
Sampson publishes on Li/O2 batteries in Nano Letters
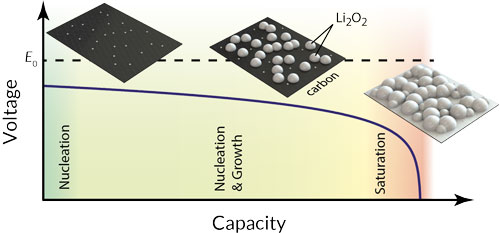
The Li–O2 battery (also known as the lithium-air battery) is an experimental class of batteries that can theoretically store three times as much energy as conventional lithium-ion batteries with the same weight. This makes it attractive for applications where lightweight energy storage is essential, such electric vehicles. However, there are a number of obstacles that must be overcome before the Li–O2 battery can be useful, one of which is its poor performance at high currents. Specifically, it has been observed that the useable capacity of the battery decreases dramatically as the current increases, and a satisfactory explanation for this has yet to be reported. Previous models of the battery suggested that the solid discharge product, lithium peroxide (Li2O2), acts as an inhibiting agent by blocking oxygen diffusion or by blocking the transport of electrons in the cathode. Neither mechanism alone was able to produce a good fit to the experimental capacity-vs-current trend, however.
Noticing that the size of lithium peroxide particles changes predictably with discharge current, Sampson proposed that the usable capacity of the battery is linked to how the particles nucleate and grow on the cathode surface. A new model was developed in which the voltage of the battery depends on the amount of exposed surface area remaining on the cathode. The electrical resistance of lithium peroxide still plays a role, but, in contrast to previous models, it does not become important until the cathode is almost completely covered by lithium peroxide. The degree of lithium peroxide coverage was calculated by deliberately tracking the nucleation and growth of all lithium peroxide particles on the cathode. The result is the first model of the Li–O2 battery that accurately predicts both the shape of the voltage profile as well as the usable capacity of the battery as a function of discharge current. This model implies that an important strategy for obtaining a high capacity out of a lithium-air battery is to promote the production of fewer, but larger, particles of lithium peroxide. Many recent experimental findings are consistent with this strategy. For example, electrolytes can be selected that dissolve lithium superoxide (LiO2), which is the precursor to lithium peroxide, to partially inhibit nucleation.
Since the paper is technically involved, Sampson wrote a blog post about it geared towards a wider audience. It can be read here: https://medium.com/@SampsonLau/connecting-the-dots-in-the-lithium-air-battery-477b91c492bc
Reference: Sampson Lau, Lynden A. Archer. “Nucleation and Growth of Lithium Peroxide in the Li–O2 Battery” Nano Letters DOI: 10.1021/acs.nanolett.5b02149
